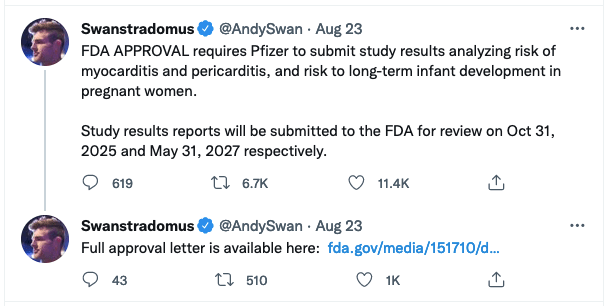
FDA APPROVAL requires Pfizer to submit study results analyzing risk of myocarditis and pericarditis, and risk to long-term infant development in pregnant women.
Study results reports will be submitted to the FDA for review on Oct 31, 2025 and May 31, 2027 respectively.
— Swanstradomus (@AndySwan) August 23, 2021
____